Review

Diaphragm Disease:Unique Small Intestinal Pathology Associated with Non-steroidal Anti-inflammatory Drugs (NSAIDs) – A Case Report with Review of Literature
1 Mona Bargotya 2 Seema Sachan 1Ankita Mehta
- 1. Rajiv Gandhi Super Speciality Hospital,Delhi,India
- 2. B.L.K Super Speciality Hospital, Delhi,India.
Submitted Thursday, May 3, 2018
Accepted Tuesday, September 18, 2018;
Published Wednesday, September 19,2018
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited
Abstract
Nonsteroidal anti-inflammatory drugs (NSAIDs) induce a spectrum of intestinal lesions in which diaphragm disease (DD) is a rare occurrence, which usually occurs after consuming these drugs for prolonged periods. Nonspecific clinical features and vague symptoms makes difficult to diagnose this disease clinically as well as by radiological investigations. Hence, it is quite difficult to differentiate DD from small intestinal tumours and inflammatory bowel disease. The diagnosis is made on final histopathology after surgical resection and anastomosis of the affected segment which is the approved treatment. Herein, we report a case of diaphragm disease presenting with sub-acute intestinal obstruction with characteristic gross and histological pathognomonic findings.
Keywords:
Diaphragm disease (DD), Nonsteroidal anti- inflammatory drugs (NSAIDs)
Introduction
Diaphragm disease (DD) is a rare gastrointestinal condition and is a relatively recent clinical entity. The range of overall prevalence of DD is broad i.e. from 8.4% to 66% [1]. Because of the vague symptoms of this disorder impeding the diagnosis, its exact prevalence is still unclear. Females are affected more than the males and the small intestine is more commonly involved than the large intestine. Presence of a single diaphragm should prompt search of others since often they are multiple. The term DD was first used by Lang
et al in 1988 describing the pathologic findings of nonspecific small bowel disease associated with the prolonged use of NSAIDs [2]. The pathogenesis of the disease is yet to be fully understood, however, it is considered characteristic of injury due to prolonged intake of NSAIDs [3].Several studies have revealed that some degree of small bowel enteropathy develops in 70% of patients with long term use of NSAIDs [4,5,6].In most of the studies it is found that the patients develop DD while taking NSAIDs, however it has been found that in occasional cases progressive damage to the bowel mucosa can occur even after the cessation of the drugs. This entity has also been reported in patients without appreciable use of NSAIDs [7].Other probable conditions that can lead to diaphragm disease are celiac disease, eosinophilic gastroenteritis, radiation injury and potassium intake [8].
DD should be considered in patients presenting with obstructive symptoms, chronic abdominal pain, nausea, vomiting, diarrhoea, anemia, weight loss and protein losing enteropathy. It is difficult to diagnose this disease on the basis of these non-specific clinical findings. In most of the cases radiological investigations also fail to differentiate it from other causes of bowel obstruction, especially small intestinal tumors, inflammatory bowel disease and tuberculosis. The ambiguity of symptoms as well as inconclusive investigations made the diagnosis complicated.
The non-invasive diagnosis of DD is challenging and most of the cases are still diagnosed on histology after surgical resection [8]. DD is characterized by short length circumferential lesions and located most commonly in the small intestine thereby leading to multiple stenosis of the lumen. There is submucosal fibrosis in the gastrointestinal tract subsequently leading to the formation of a stricture resembling diaphragm. The diaphragms are commonly multiple and rarely single. They appear as a tissue disk which is protruding into the lumen of the intestine circumferentially, thereby reducing the lumen of the small intestine to a smaller diameter. Histologically, the diaphragm comprises of mucosa, submucosa without involvement of muscularis propria and serosa.
Herein, a case is presented with discussion on etiology, pathogenesis, diagnosis and treatment along with review of available literature.
Case Report
A 52 years old female presented with gradually worsening chronic colicky abdominal pain and abdominal distension of three months duration. The pain was usually followed by vomiting. No diagnostic findings were revealed on extensive preoperative endoscopic and radiological workup. Laboratory tests were also unremarkable. Retrospective relevant history of long term intake of NSAIDs (aspirin and diclofenac) for chronic joint pain was present however, exact duration couldn’t be specified.
Patient underwent a diagnostic laparotomy in view of deteriorating condition. Per- operatively multiple strictures were identified in the ileum with dilatation of the proximal jejunum and ileum. However, no palpable mass was identified grossly. Resection of the effected segment of ileum was done followed by anastomosis. Segment of small intestine was received in the Department of Pathology with accompanying clinical notes of multiple strictures in the ileum. On gross examination (Figures 1 , 2A & 2B), the intact segment of small intestine measured 18 cm in length with overall bland serosal aspect showing mild vague thickenings with ring like depressions at irregular intervals throughout its length. Mucosal aspect showed segmentation of intestine by multiple, unevenly distributed, incomplete mucosal diaphragms resembling perforated diaphragms with reduction of lumen size to 5-8 mm. The intestinal compartments showed focal haemorrhagic roughened areas. Bulk of the mucosa appeared normal.
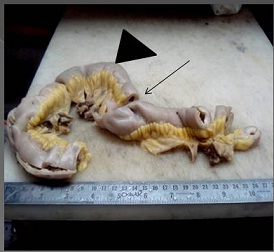

Figure 1: Bland serosal aspect of resected ileum with ring like depressions on external surface (arrow heads), difficult to appreciate the lesion seen (arrow) before the bowel is opened.

Figure 2A &2B :Incomplete mucosal diaphragms resembling perforated diaphragms (End on appearance).
Microscopically ((Figures 3 4 & 5), the diaphragm comprised of mucosa and submucosa composed of sclerosed eosinophilic connective tissue in the submucosa intermixed with smooth muscle, nerve fibres and vessels associated with variable mild to moderate chronic lymphoplasmacytic infiltrate admixed with few eosinophils. The smooth muscle component was represented by chaotically arranged muscle bundles. No granulomas were seen. There was no evidence of dysplasia. The gross and microscopic findings were consistent with classical picture of DD.

Figure 3:Low power view of diaphragm lesion showing widened submucosa

Figure 4: Scanner view of widened submucosa composed of sclerosed eosinophilic connective tissue and highlights choatically arranged smooth muscle bundles

Figure 5: High power of submucosal lesion highlighting inflammation in addition to haphazard arrangement of smooth muscle bundles
Postoperative recovery was reported to be uneventful and the patient was discharged after 7 days. After one year of regular visits, the patient was lost to follow up.
Discussion
NSAIDs are the most commonly used analgesic as well anti- inflammatory drugs in clinical practice today. It is well known that NSAIDs cause a high incidence of toxic effects on the mucosa of the gastrointestinal tract, damaging the walls of the stomach and duodenum along with causing enteropathy of the small bowel. Dyspepsia, gastric ulceration, erosions and bleeding are secondary to mucosal injury.
DD is one of the NSAIDs induced enteropathies. NSAIDs are thought to inhibit cyclooxygenase -1 which results in reduced microcirculatory blood flow leading to localized ischemia, ulcers, strictures and ultimately mucosal diaphragms [2,8-13].Modulation of signal transduction pathways such as nuclear factor- κβ and peroxisome proliferator- activated receptor γ, increasing intestinal permeability and uncoupling of mitochondrial oxidative phosphorylation are the other proposed mechanisms which may work alone or in combination to decrease leucocyte migration and adhesion hence facilitating invasion by toxins and bacteria. However, the exact pathogenesis of this entity has yet not been elucidated thus far, it is almost certainly multifactorial. High doses taken daily generally are also supposed to be associated with DD. The pathophysiology of NSAIDs-induced mucosal injury may be different in different parts of the gastrointestinal tract.
Recent studies have revealed that in addition to DD, NSAIDs- related injury should be considered in villous atrophy, eosinophilic enteritis, inflammatory fibroid polyp, neuromuscular and vascular hamartoma (NMVH) and in a recently added entity called NSAID associated submucosal fibrous nodules of small intestine.
Radiological diagnosis may fail to characterize the disease and hence most of the cases are still diagnosed on histology after surgical resection [14]. The diaphragms are commonly characterized histologically by typical multiple mucosal projections, mucosal ulcerations, villous atrophy, increased inflammatory cells, submucosal fibrosis, chaotic arrangement of smooth muscle fibres, presence of vascular and neural elements along with serosal constrictions resulting in luminal obstruction. Recent imaging modalities like capsule endoscopy, double balloon enteroscopy and combined enteroscopy with enterocylsis may help in the confirmation of the diagnosis as well as to delineate the extent of resection [15].
Conclusion
Although rarely reported in literature, increasing use as well as recognition of the possible adverse effects of NSAIDs is making the diagnosis of DD more common than previously considered. Diagnosis is challenging hence awareness with a high index of suspicion followed by extensive workup is necessary. DD should be considered in the differential diagnosis of the patients presenting with long term history of NSAIDs, GI tract bleeding, anemia and chronic abdominal pain with or without evidence of bowel obstruction. The definitive diagnosis is made intra operatively in most of the cases followed by histopathological examination. Laparotomy and small bowel resection is the treatment of choice because of the low yield of the standard radiologic and endoscopic methods.
References
[1]Kelly ME, McMahon LE, Jaroszewski DE, Yousfi MM, Petris GD, Swain JM. Small-Bowel Diaphragm Disease Seven Surgical Cases. ARCH SURG 2005; 140:1162-66.[PubMed]
[2]Lang J, Price AB, Levi AJ, Burke M, Gumpel JM, Bjarnason I. Diaphragm disease: pathology of disease of the small intestine induced by non-steroidal antiinflammatory drugs. J ClinPathol. 1988;41:516-26.[PubMed][PMC Full Text]
[3]Lee FD. Drug related pathological lesions of the intestinal tract. Histopathology. 1994;23:303-308.[PubMed]
[4]Bjarnason I, Hayllar J, MacPherson AJ, Russell AS. Side effects of nonsteroidal anti-inflammatory drugs on the small and large intestine in humans. Gastroenterology 1993; 104:1832–18470.[PubMed]
[5]Pilgrim S, Velchuru V, Waters G, Tsiamis A, Lal R. Diaphragm disease and small bowel enteropathy due to nonsteroidal anti-inflammatory drugs: a surgical perspective. Colorectal Dis 2011; 13:463–466.[PubMed]
[6]Velchuru VR, Rimal D, Studley JG, Sturzaker HG. Diaphragm disease of the ileum. Int J Surg 2006; 4:184–186.[PubMed]
[7]Santolaria S, Cabezali R, Ortego J, Castiela T, Salinas JC,Lanas A.Diaphragm disease of the small bowel: a case without apparent nonsteroidal antiinflammatory drug use. J ClinGastroenterol. 2001; 32:344-346.[PubMed]
[8]De Petris G, Lopez JI. Histopathology of diaphragm disease of small intestine: a study of 10 cases from a single institution. Am J ClinPathol2008;130:518-25.[PubMed]
[9]Levi S, de Lacey G, Price AB, Gumpel MJ, Levi AJ, Bjarnason I. Diaphragm like strictures of the small bowel in patients treated with non-steroidal antiinflammatory drugs. Br J Radiol1990;63:186-89.[PubMed]
[10]McCune KH, Allen D, Cranley B. Small bowel diaphragm disease: stricture associated with non-steroidal anti-inflammatory drugs. Ulster Med J 1992;61:182-184.[PubMed] [PMC Full Text]
[11Shumaker DA, Bladen K, Katon RM. NSAID-induced small bowel diaphragms and strictures diagnosed with intraoperative enteroscopy. Can J Gastroenterol 2001; 15:619–623.[PubMed]
[12]Slesser AA, Wharton R, Smith GV,Buchanan GN. Systematic review of small bowel diaphragm disease requiring surgery. Colorectal Dis 2012; 14:804–813.[PubMed]
[13]Bjarnason I, Takeuchi K Intestinal permeability in the pathogenesis of NSAID induced enteropathy J. Gastroenterol 2009; 44(suppl 19):23–29.[PubMed]
[14]Ullah S, Ajab S, Rao R, Raghunathan G, DaCosta P. Diaphragm Disease of the Small Intestine:An Interesting Case Report. International Journal of Surgical Pathology 2015;23(4):322–24. [PubMed]
[15]Hayashi Y, Yamamoto H, Taguchi H, et al. Nonsteroidal anti-inflammatory drug induced small-bowel lesions identified by double-balloon endoscopy: endoscopic features of the lesions and endoscopic treatments for diaphragm disease. J Gastroenterol. 2009;44.[PubMed]

